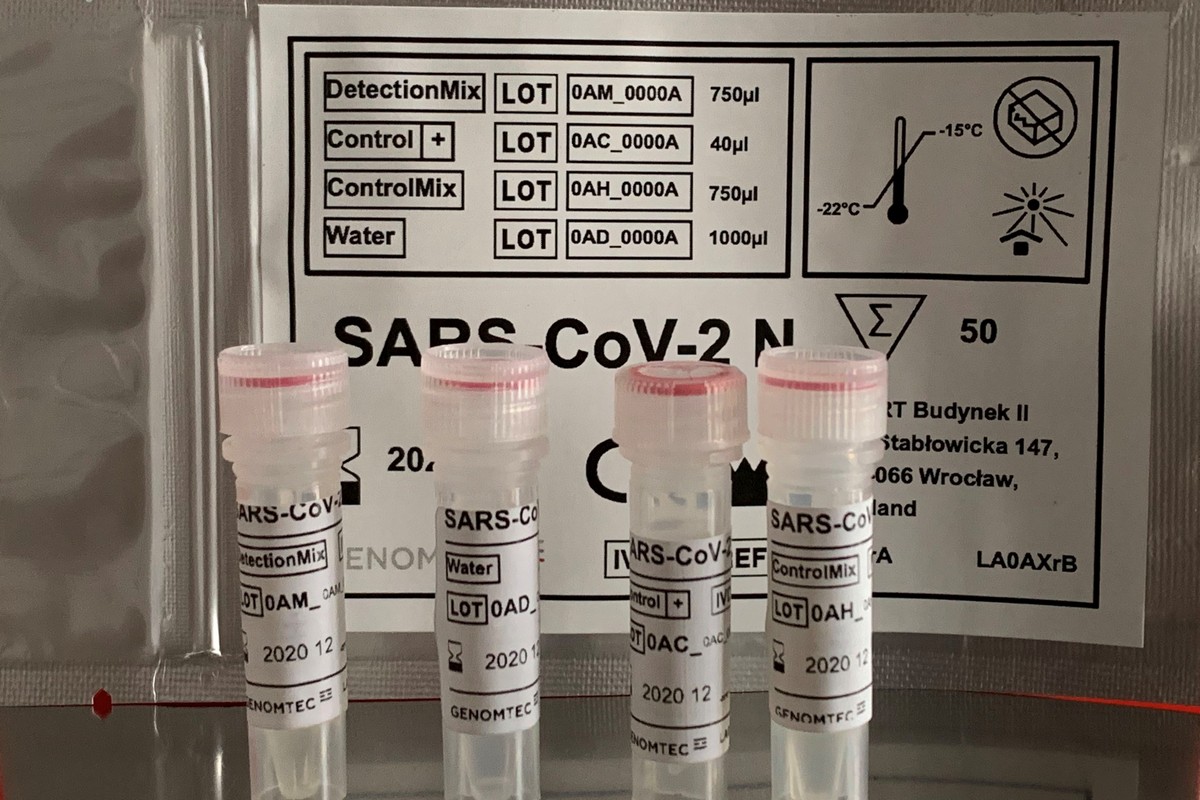
Zestaw diagnostyczny Genomtec® SARS-CoV-2 RT-LAMP / N CE-IVD to nasz pierwszy produkt przeznaczony do diagnostyki COVID-19. Po nieco ponad 10 tygodniach ciężkiej pracy jesteśmy niezmiernie dumni, że możemy pomóc w usprawnieniu pracy laboratoriów, udostępniając nasz szybki test genetyczny, który wykorzystuje technologię SNAAT®, zapewniając tym samym doskonałe parametry diagnostyczne i spełniając długofalową obietnicę Genomtec: nasze testy są nieporównywalnie szybsze od innych dostępnych na rynku zestawów diagnostycznych w technice RT-PCR.
Osiągnięte parametry wynikają z zastosowania naszych zgłoszonych do opatentowania starterów LAMP, najwyższej jakości enzymów i zoptymalizowanej kompozycji odczynników. Wszystko to gwarantuje wykrycie sygnału dla pozytywnych próbek klinicznych w mniej niż 20 min., przy maksymalnym czasie reakcji wynoszącym 30 minut (w porównaniu do minimum 75 minut dla najszybszych laboratoryjnych testów RT-PCR). Ponadto został on klinicznie zweryfikowany na próbkach pacjentów ze 100% zgodnością względem testów RT-PCR CE-IVD i jest dostępny do użycia na dowolnym termocyklerze Real-Time wyposażonym w detekcję fluorescencji w kanale FAM (zielony).
Obecnie zainteresowanie naszym testem wyraził szereg krajów w wielu rejonach świata. Jednak nie zatrzymujemy się tutaj – będziemy dalej rozwijać nasze portfolio testów w kierunku SARS-CoV-2, aby zaoferować wkrótce nowe, innowacyjne rozwiązania diagnostyczne, jednocześnie pracując nad naszą mobilną platformę POCT – Genomtec ID.